What is Superfluid?
A superfluid is a phase of matter capable of flowing endlessly without energy loss. This property of certain isotopes was discovered by Pyotr Leonidovich Kapitsa, John F. Allen, and Don Misener in 1937. It has been achieved at very low temperatures with at least two isotopes of helium, one isotope of rubidium, and one isotope of lithium.
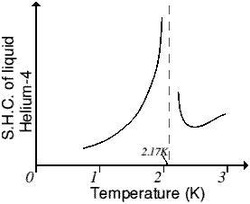
A superfluid can be a liquid or a gas, but not a solid. For example, helium's freezing point is 1K (Kelvin) and 25 atmospheres of pressure, the lowest of any element, but the substance begins exhibiting superfluid properties at about 2K. The superfluid phase transition occurs when all the constituent atoms of a sample begin to occupy the same quantum state. This point is called as Lambda point. It is called lambda point because, the graph of specific heat capacity of Helium-Temperature graph represents a Greek letter Lambda.This transition happens when the atoms are placed very closely together and cooled down so much that their quantum wave functions begin to overlap and the atoms lose their individual identities, behaving more like a single super-atom than an agglomeration of atoms.
A limiting factor on which materials can exhibit superfluidity and which cannot is that the material must be very very cold (<4K) and remain fluid at this cold temperature. Materials which become solid at low temperatures cannot become superfluids. When cooled to very low temperatures, a superfluid-ready set of bosons, atoms with an even number of nucleons, forms into a Bose-Einstein condensate, a superfluid phase of matter. When fermions, atoms with an odd number of nucleons such as the helium-3 isotope, are cooled down to a few Kelvin, this is not sufficient to cause a superfluid transition.
A superfluid can be a liquid or a gas, but not a solid. For example, helium's freezing point is 1K (Kelvin) and 25 atmospheres of pressure, the lowest of any element, but the substance begins exhibiting superfluid properties at about 2K. The superfluid phase transition occurs when all the constituent atoms of a sample begin to occupy the same quantum state. This point is called as Lambda point. It is called lambda point because, the graph of specific heat capacity of Helium-Temperature graph represents a Greek letter Lambda.This transition happens when the atoms are placed very closely together and cooled down so much that their quantum wave functions begin to overlap and the atoms lose their individual identities, behaving more like a single super-atom than an agglomeration of atoms.
A limiting factor on which materials can exhibit superfluidity and which cannot is that the material must be very very cold (<4K) and remain fluid at this cold temperature. Materials which become solid at low temperatures cannot become superfluids. When cooled to very low temperatures, a superfluid-ready set of bosons, atoms with an even number of nucleons, forms into a Bose-Einstein condensate, a superfluid phase of matter. When fermions, atoms with an odd number of nucleons such as the helium-3 isotope, are cooled down to a few Kelvin, this is not sufficient to cause a superfluid transition.
